BIOTRONIK Sets New Clinical Benchmark with Ultrathin Strut Orsiro Coronary Drug-Eluting Stent Final 5-Year Data from BIOFLOW-V Trial Confirms Better Safety for Orsiro DES, Confirming Orsiro’s Position as the Clinical Benchmark for DES
BÜLACH, Switzerland, February 28, 2022 – BIOTRONIK is pleased to announce five-year data from the BIOFLOW-V trial, which was presented yesterday at the 2022 CRT Conference during a late-breaking clinical trial session by Dr. David Kandzari, US Principal Investigator, Piedmont Heart Institute, Atlanta, Georgia.
The five-year follow-up data demonstrates consistently lower clinical event rates in target lesion failure (TLF) and significantly lower rates of target-vessel myocardial infarction (TV-MI). This reinforces the body of evidence differentiating Orsiro® DES from other contemporary DES making it the first and only ultrathin strut DES proven to outperform Xience DES.1,2 Orsiro DES received FDA approval in 2019 and has been used to treat more than three million patients worldwide to date. The latest generation of BIOTRONIK’s DES, Orsiro® Mission, gained FDA approval in 2021 and benefits from the same stent as its predecessor device with a re-engineered delivery system for even better deliverability*.
“The final five-year follow-up data confirms Orsiro’s superior safety profile and low TLF rates,” said Dr. David Kandzari. “The ultrathin strut Orsiro stent significantly reduces the risk for TV-MI and shows a low rate of stent thrombosis (ST), and specifically a significant reduction in late and very late stent thrombosis. These long-term results confirm that the Orsiro DES sets a new clinical benchmark in PCI treatment options.”
Orsiro’s clinical performance in the pivotal BIOFLOW-V trial (n=1,334)2 has already demonstrated significantly lower rates for TLF and TV-MI compared to Xience DES at 12 months follow-up and continued to show significant differences across the most important secondary endpoints at 36 months follow-up.3
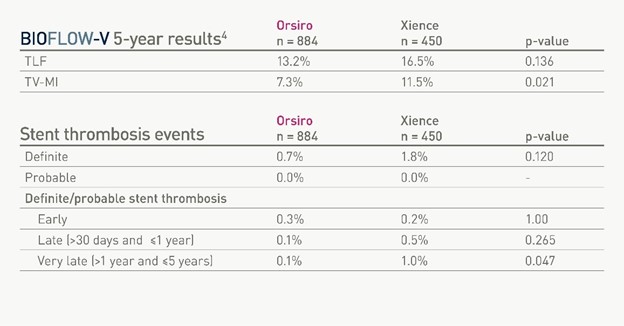
“The final BIOFLOW-V data clearly shows that Orsiro provides better patient outcomes in this head-to-head comparison against Xience DES”, said Ryan Walters, President at BIOTRONIK, Inc. “With the newly launched Orsiro Mission DES available in the U.S. we have the same clinical performance with next level deliverability making it a 'must have' in every cath-lab."
BIOFLOW-V is the first large randomized trial to show improved long-term outcomes in the field of DES over Xience DES. The international study included 1,334 patients, of which a sizable percentage were complex: 50.8%5 of patients presented with acute coronary syndrome (ACS), 35.0%5 of patients were diabetic, and 73.8%6 of lesions were ACC/AHA Class B2/C.
-END-
*Clinical data conducted with Orsiro, Orsiro Mission’s predecessor device can be used to illustrate Orsiro Mission clinical data.
**BIOTRONIK data on file, status as per January 2020
Orsiro and Orsiro Mission are trademarks or registered trademarks of the BIOTRONIK Group of Companies.
References:
1 Xience is a trademark or registered trademark of the Abbott Group of Companies.
2 Kandzari D et al. The Lancet. 2017, 390(10105).
3 Kandzari D et al. J Am Coll Cardiol. Cardiovasc Interven. 2020, doi: 10.1016/j.jcin.2020.02.019.
4 Kandzari D presented at CRT 2022 – Late Breaking Clinical Trial February 27th, 2022, Late Breaking Trials: Session I
5 Kandzari et al, Supplement to: Kandzari DE, Mauri L, Koolen JJ et al. Ultrathin Bioresorbable Polymer Sirolimus-Eluting Stents versus Thin Durable Polymer Everolimus-Eluting Stents in Patients Undergoing Coronary Revascularisation (BIOFLOW V): a randomised trial, JACC, 2017
6 Kandzari, BIOFLOW V – Comparison of ultrathin sirolimus eluting bioresorbable polymer with thin everolimus eluting durable polymer stents, Presented at ESC Congress, Barcelona, 2017
For indications please see Instructions For Use.
About BIOTRONIK:
At BIOTRONIK, patient well-being is our top priority and has been for 60 years. BIOTRONIK is a leading global medical technology company with products and services that save and improve the lives of millions suffering from heart and blood vessel diseases as well as chronic pain. Driven by a purpose to perfectly match technology with the human body, we are dedicated innovators who develop trusted cardiovascular, endovascular and neuromodulation solutions. BIOTRONIK is headquartered in Berlin, Germany, and is represented in over 100 countries.